


Acids, Bases, and the pH Scale – Interactive Google Slides Lesson + Alien Juice Bar Virtual Lab
Drag-and-Drop Simulation, Indicators, Litmus Testing, and pH Interpretation Practice.
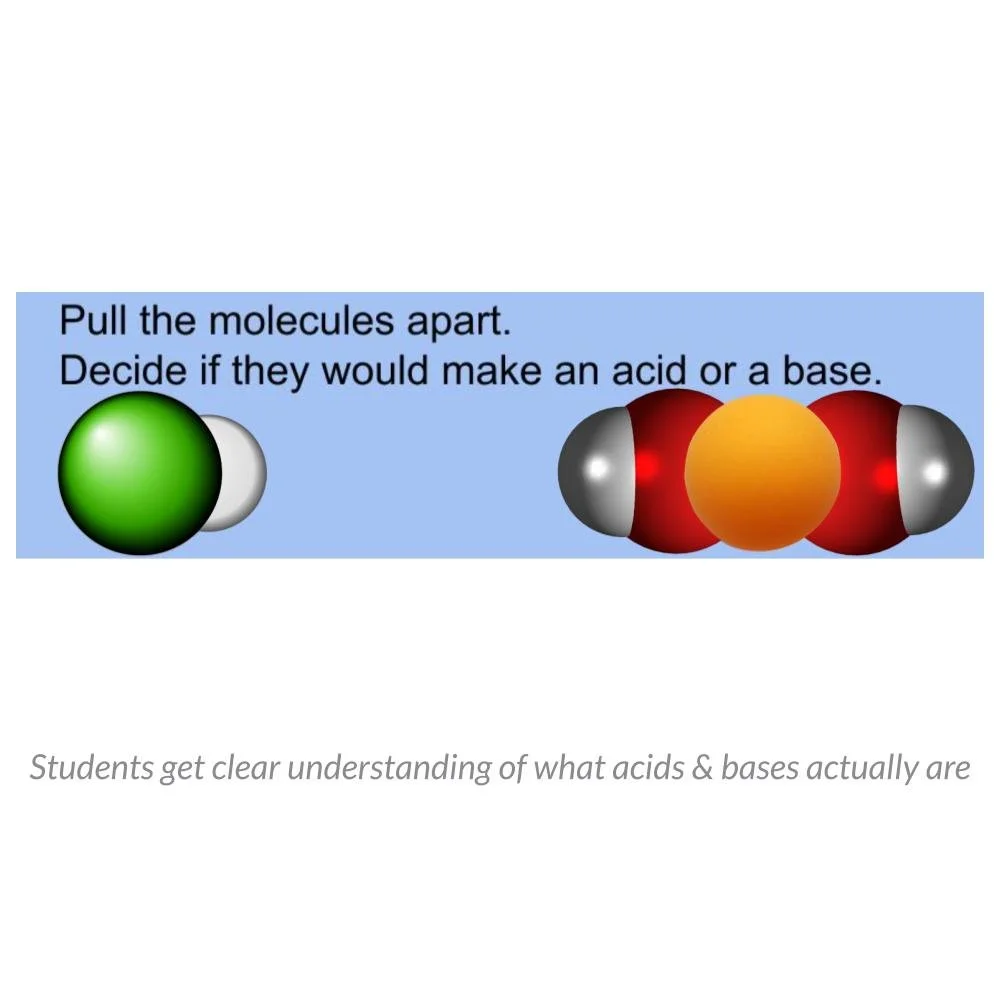
This interactive digital lesson introduces students to acids, bases, and the pH scale through molecular modeling and structured application tasks. Students work directly with particle-level representations to understand how hydrogen and hydroxide ions determine whether a substance is acidic, basic, or neutral.
The lesson is designed to move from conceptual foundations to quantitative reasoning and then to application, making it suitable for middle school and early high school chemistry or physical science courses.
What Students Do
Students begin by modeling the dissociation of water and other molecules to observe the formation of:
• hydrogen ions (H⁺)
• hydroxide ions (OH⁻)
They classify molecules based on ion production and determine whether each substance is acidic or basic.
Next, students analyze the pH scale and interpret its logarithmic structure, answering guided questions and placing values and images on a visual model of the scale.
Finally, students apply their understanding through a set of practice tasks that require them to:
• interpret pH values
• compare relative acidity and basicity
• connect molecular structure to pH behavior
Key Concepts Reinforced
• Acids vs. bases at the molecular level
• Hydrogen and hydroxide ions
• Neutral solutions
• The pH scale and logarithmic change
• Particle-level reasoning
• Conceptual chemistry modeling
Why Teachers Use This Lesson
• Visualizes abstract chemistry concepts
• Connects molecular structure to pH values
• Reinforces quantitative reasoning
• Works well as guided practice or independent work
• Suitable for digital classrooms
• Minimal prep required
Format
This lesson is delivered as a digital Google Slides activity in which students interact directly with diagrams and models.
A teacher key is included.
Best Fit For
• Middle school chemistry
• High school chemistry (introductory units)
• Physical science
• Units on acids, bases, or pH
• Classrooms introducing molecular explanations
To see a preview of this lesson, click here
NGSS Alignment (Middle School):
MS-PS1-2
NGSS Alignment (High School):
HS-PS1-3
Science & Engineering Practices (SEPs):
Analyzing and Interpreting Data; Planning and Carrying Out Investigations; Developing and Using Models
Crosscutting Concepts (CCCs):
Cause and Effect; Patterns; Structure and Properties of Matter
Common Core (Literacy in Science):
RST.6-8.3, RST.9-10.3
RST.6-8.4, RST.9-10.4
RST.6-8.7, RST.9-10.7
Daily slide + literacy - based exit ticket included with purchase
Join the Lesson Laboratory and Teach for Tomorrow!
Drag-and-Drop Simulation, Indicators, Litmus Testing, and pH Interpretation Practice.
This interactive digital lesson introduces students to acids, bases, and the pH scale through molecular modeling and structured application tasks. Students work directly with particle-level representations to understand how hydrogen and hydroxide ions determine whether a substance is acidic, basic, or neutral.
The lesson is designed to move from conceptual foundations to quantitative reasoning and then to application, making it suitable for middle school and early high school chemistry or physical science courses.
What Students Do
Students begin by modeling the dissociation of water and other molecules to observe the formation of:
• hydrogen ions (H⁺)
• hydroxide ions (OH⁻)
They classify molecules based on ion production and determine whether each substance is acidic or basic.
Next, students analyze the pH scale and interpret its logarithmic structure, answering guided questions and placing values and images on a visual model of the scale.
Finally, students apply their understanding through a set of practice tasks that require them to:
• interpret pH values
• compare relative acidity and basicity
• connect molecular structure to pH behavior
Key Concepts Reinforced
• Acids vs. bases at the molecular level
• Hydrogen and hydroxide ions
• Neutral solutions
• The pH scale and logarithmic change
• Particle-level reasoning
• Conceptual chemistry modeling
Why Teachers Use This Lesson
• Visualizes abstract chemistry concepts
• Connects molecular structure to pH values
• Reinforces quantitative reasoning
• Works well as guided practice or independent work
• Suitable for digital classrooms
• Minimal prep required
Format
This lesson is delivered as a digital Google Slides activity in which students interact directly with diagrams and models.
A teacher key is included.
Best Fit For
• Middle school chemistry
• High school chemistry (introductory units)
• Physical science
• Units on acids, bases, or pH
• Classrooms introducing molecular explanations
To see a preview of this lesson, click here
NGSS Alignment (Middle School):
MS-PS1-2
NGSS Alignment (High School):
HS-PS1-3
Science & Engineering Practices (SEPs):
Analyzing and Interpreting Data; Planning and Carrying Out Investigations; Developing and Using Models
Crosscutting Concepts (CCCs):
Cause and Effect; Patterns; Structure and Properties of Matter
Common Core (Literacy in Science):
RST.6-8.3, RST.9-10.3
RST.6-8.4, RST.9-10.4
RST.6-8.7, RST.9-10.7
Daily slide + literacy - based exit ticket included with purchase